Tulio Caldonazo1, Mateo Marin-Cuartas2, Hristo Kirov1, Torsten Doenst1
1Department of Cardiothoracic Surgery, Jena University Hospital, Friedrich Schiller University of Jena, Jena, Germany
2University Department of Cardiac Surgery, Leipzig Heart Center, Leipzig, Germany
Address for correspondence:
Torsten Doenst, MD, PhD
Department of Cardiothoracic Surgery
University of Jena
Am Klinikum 1
07747 Jena, Germany
Tel.: +49 3641 932 2901
Fax: +49 3641 932 2902
Email: doenst@med.uni-jena.de
English
The 2022 joint ESC/EACTS review of the recommendations on left main coronary artery disease in the 2018 myocardial revascularization guidelines focuses on individualizing treatment based on disease mechanisms and complexity. In general, surgical revascularization is recommended in more complex patients with a higher risk of myocardial infarction since it provides greater protection against future myocardial infarction. Patients with less complex left main disease have the option of surgical or percutaneous revascularization, considering the risk of myocardial infarction and adverse events. The review highlights the importance of individualized decision-making for patients with left main disease, given the broad spectrum of cases, from isolated ostial lesions to extensive coronary disease. The elimination of the SYNTAX Score calculation is justified, considering the lethality of the left main occlusion. The recommendation of percutaneous revascularization for proximal/ostial lesions without significant additional coronary disease aims to reduce invasiveness, while surgical revascularization is preferred for distal lesions and complex coronary disease. In summary, the 2022 joint ESC/EACTS review of the recommendations on left main coronary artery disease in the 2018 myocardial revascularization guidelines reflects available data in the literature and supports individualization of treatment based on disease mechanisms and complexity. This mechanistic approach helps understand the need to promote personalized decisions focused on each individual patient.
Español
La reciente revisión conjunta ESC/EACTS de 2022 de las recomendaciones sobre enfermedad de tronco izquieroa en las guias de revascularización miocárdica del 2018 se centra en individualizar el tratamiento según los mecanismos y la complejidad de la enfermedad. A los pacientes más complejos, con mayor riesgo de infarto, generalmente se les recomienda la revascularización quirúrgica, que proporciona una mayor protección contra futuros infartos miocárdios. Los pacientes con una enfermedad menos compleja cuentan con la opción de revascularización quirúrgica o percutánea, teniendo en cuenta el riesgo de infarto miocardio y eventos adversos.La revisión destaca la importancia de la toma de decisiones individualizada para los pacientes con enfermedad coronaria de tronco izquierdo, dado el amplio espectro de presentación, desde lesiones ostiales aisladas hasta enfermedad coronaria extensa. Se justifica la eliminación del cálculo del SYNTAX Score, considerando la letalidad de la oclusión del tronco izquierdo. La recomendación de revascularización percutánea para lesiones proximales/ostiales sin enfermedad coronaria significativa de otros vasos tiene como objetivo reducir la invasividad, mientras que en las lesiones distales y la enfermedad coronaria compleja se prefiere la revascularización quirúrgica. En resumen, la reciente revision de las guias reflejan los datos disponibles en la literatura y respaldan la individualización del tratamiento según los mecanismos y la complejidad de la enfermedad. Este enfoque mecanístico ayuda a comprender la necesidad de promover decisiones personalizadas centradas en cada paciente.
Português
A avaliação das diretrizes e da revisão sobre doença do tronco da coronária esquerda (TCE) enfoca a individualização do tratamento com base em mecanismos e complexidade da doença. Pacientes mais complexos, com maior risco de infarto, geralmente recebem recomendação de revascularização cirúrgica, que fornece maior proteção contra futuros infartos. Pacientes com doença menos complexa têm opções de revascularização cirúrgica ou percutânea, levando em consideração o risco de infarto e eventos adversos invasivos.A revisão ressalta a importância da decisão individualizada para pacientes com doença do TCE, dado o amplo espectro de casos, desde lesões ostiais isoladas até doença coronariana extensa. A eliminação do cálculo do SYNTAX Score é justificada, considerando a letalidade da oclusão do TCE. A recomendação de revascularização percutânea para lesões proximais/ostiais sem doença coronariana significativa de outros vasos visa reduzir a invasividade, enquanto lesões distais e doença coronariana complexa recebem preferência por revascularização cirúrgica. Em resumo, as diretrizes atuais e a revisão da doença do TCE refletem os dados disponíveis na literatura e apoiam a individualização do tratamento com base em mecanismos e complexidade da doença. Esta abordagem mecanicista ajuda a compreender a necessidade de promover decisões personalizadas e focadas em cada paciente.
Click here to access the 2022 joint ESC/EACTS review of the recommendations on left main coronary artery disease in the 2018 myocardial revascularization guidelines
Start with “why” (Simon Sinek)
Cardiac surgery is thinking with your hands (Torsten Doenst)
According to prominent publications in the field of management and leadership, successful business organizations share the feature that their employees know “why” they are doing their work1. According to that perspective, workers knowing what and how to do their jobs may perform reasonably, but performance reaches another level once employees understand the purpose of their work, which is reflected in the little word “why”. We published a surgical perspective on a similar issue several years ago2, where we apply the same logic to the field of cardiac surgery, reflected in our motto: “cardiac surgery is thinking with your hands”. This motto suggests that knowing “why” you do “what” and “when” in cardiac surgery (i.e., in- and outside of the operating room as well as in clinical decision-making) reduces complications, increases performance, and improves patient outcomes2.
In medicine, asking “why” generally reflects investigating the underlying mechanisms. This can be impressively illustrated in the field of pharmacology, where treatment effects can often be explained by specific molecular interactions of drugs with an intracellular target. For instance, statins inhibit a specific enzyme - HMG-CoA reductase - responsible for cholesterol synthesis. Mechanistic investigations often require animal research because interventions to demonstrate true causality are not possible in humans. Thus, epidemiological methods have been developed to assess complex treatment effects in humans3, 4. Thus, expert committees generally evaluate observational evidence and provide recommendations in form of guidelines by weighing outcomes from primarily randomized but also non-randomized evidence. Interestingly, these investigations are almost never driven by efforts to explore the “why” (i.e., the underlying mechanisms) but generally explore the “what” and “how” (e.g., “what” is better – percutaneous coronary intervention (PCI) or coronary artery bypass grafting (CABG)? “How” is PCI or CABG performed best, etc.). As a consequence, the ability of guideline committees to mechanistically explain differences is limited. The currently available European guidelines for the invasive treatment of coronary artery disease (CAD)5 and the 2022 review of the left main (LM) recommendations6, as official statements of the European societies (European Society of Cardiology – ESC, and European Association of Cardio-Thoracic Surgery- EACTS), can therefore only provide guidance about “what” to do in individual patients and partially even “how” to do it best. However, guidelines experience inherent difficulties in deducting the “why” from the available data. In this Editorial, we attempt to provide a “why” for the treatment of coronary artery disease (CAD) with CABG, PCI, or medical therapy and put the guideline recommendations and their recent review of LM disease recommendations into perspective. We discuss a treatment mechanism that has the potential to explain most current controversies, and that may enhance our ability to care for patients with chronic CAD, including those with LM disease.
Why should “revascularization” treatments differ in treatment effects?
If both PCI and CABG “revascularize” ischemic myocardium, why do we expect differences in treatment effects? It appears to be a common notion that chronic obstructive CAD causes myocardial ischemia and that alleviation of obstructions is responsible for the observed treatment effect5,7. This understanding finds support by discussions that complete revascularization is better than incomplete revascularization, which is led independently of the treatment modality chosen8-10. It is further supported by our systematic attempts to quantify the amount of ischemic myocardium in the patient’s heart11-14. Despite the general acceptance of this pathophysiological understanding and the use of the current terminology over decades, there are open questions that have been repetitively brought up by investigations with controversial results15-17. For instance, why is testing for ischemia unable to predict a revascularization treatment effect18? Why has there never been an improvement in life expectancy with PCI in chronic coronary syndrome in randomized trials19, 20. Why is there repeated evidence in chronic coronary syndrome for CABG prolonging life versus medical therapy or PCI20 and why is there no difference between PCI and CABG in patients with lower anatomic CAD complexity9, 21? Finally, why is there no increase in ejection fraction with revascularization in patients with chronic CAD and left ventricular dysfunction22, 23? Assessing the observed treatment effects and reviewing our current understanding of CAD pathophysiology, it appears necessary to reconsider our mechanistic understanding of CAD pathology and the invasive treatment options.
Mechanistic thinking can resolve controversies
Based on the currently available data, one may argue that if treating chronic obstructive CAD with PCI is not associated with improvement in survival20, inducible ischemia may not limit life expectancy. If it still does, this effect would not be modified with PCI. We suggested that any life-prolonging effect of invasive CAD treatment in chronic CAD is related to the prevention of myocardial infarctions24. Since the majority of infarctions arise from coronary lesions that are non-obstructive, addressing obstructive lesions alone may successfully treat symptoms from inducible ischemia, but are much less able to prevent myocardial infarctions. Therefore, placing a bypass graft generally distal to all chronic stenotic coronary lesions provides a surgical collateral, explaining the infarct-preventative effect of CABG. PCI, in contrast, treats flow-relevant lesions only, provides the same symptomatic relief as CABG25, but can only prevent a small number of future myocardial infarctions18, 26. Since not all myocardial infarctions are lethal, it becomes much more difficult to demonstrate a survival impact with PCI than with CABG. This concept unifies presumably contradictory data and has the potential to resolve all current controversies15. In patients with low risk of myocardial infarction (as is likely the case in less anatomically complex CAD), CABG and PCI can present similar results. This recognition also suggests that a life-prolonging impact of both treatments may be small or even absent under these conditions. If CAD is complex and risk of future myocardial infarctions is high, the overall risk of dying from infarctions is higher and CABG is superior by preventing myocardial infarctions also from non-flow limiting lesions and therefore has an advantage over PCI or medical therapy in chronic CAD. If this concept is accepted, patients may benefit from a mechanistic approach focusing on assessing infarction risks in chronic CAD. It also means, that using the term “revascularization” for both PCI and CABG alike is inappropriate because it does not reflect the individual treatment mechanism required for the individual patient15. The surgical collateralization effect of CABG may not be the first thing that comes to mind with “revasculaization” but is the most important mechanism of action for the prognosis of patients with complex chronic CAD.
Does the mechanistic concept of infarct-prevention also apply to left main disease?
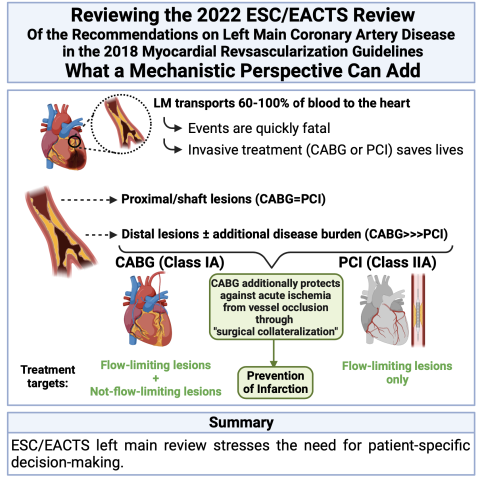
Applying this concept to patients with LM disease is equally able to explain the results. LM disease is special because it is quickly associated with mortal adverse events. Our daily language reflects this risk. LM PCI without the presence of a patent bypass graft to the left system is termed “unprotected”27. Morever, the LM is also known as “the widow maker”. Such statements are not made for technically sometimes even more demanding interventions on one individual vessel (e.g., in case of chronic total occlusions - CTO)28. Indeed, a severe LM stenosis is related to limitations in blood supply to a large part of cardiac muscle, so any disruption in blood flow can cause a significant and immediate reduction in cardiac output, heart failure, arrhythmias, or sudden cardiac death29. Natural mortality risk of patients with LM disease is highest among CAD patients29. As a consequence, the early demonstration of a survival advantage of CABG compared to medical therapy was accepted as proof for the prognostic treatment effect of invasive treatment30. As a consequence, trials comparing PCI to medical therapy in patients with LM have never been performed. Thus, in patients with LM disease (>50% stenosis), independent of the anatomic complexity of the remaining coronary tree, it is assumed that invasive treatment prolongs life, even though that has never been specifically demonstrated in randomized trials for PCI31. From our mechanistic, infarction-prevention perspective this notion is plausible, because the occlusion of the LM is a lethal event in the vast majority of cases. Thus, mechanically addressing the obstruction in the LM has a prognostic potential, which (specifically in cases with low CAD in the remaining vessels – meaning low SYNTAX score) should be equally be exploitable by CABG and PCI. Since distal LM disease is most frequently associated with more complex disease32 and is associated with significantly more procedure-related complications and poorer long-term effects with PCI10, 33, CABG should outperform PCI in those cases. Indeed, CABG appears to outperform PCI in patients with distal LM in almost all outcomes (including mortality) while there is no difference for proximal or shaft lesion, according to a recent meta-analysis34. In addition, the most recent patient-level meta-analysis on all LM trials (most of the patients were of lower anatomic complexity) was not able to demonstrate a clear reduction in mortality with CABG compared to PCI but there was a highly significant reduction in the occurrence of future myocardial infarctions26, which lends further support to the collateralization hypothesis15 even in patients with LM disease. All these considerations also suggest that treatment strategies must be developed specifically for every individual case, because the heterogeneity of characteristics in patients presenting with LM disease may be enormous32.
Does the mechanistic perspective fit to the guidelines and the left main review?
As mentioned above, for methodological reasons, guidelines are bound to the observations presented in clinical trials (preferably randomized). Assessing the guidelines5 and the LM review6 from our mechanistic perspective15 reveals a very good match. More complex patients (who are likely to have a greater infarction risk) receive primarily a CABG treatment recommendation (where surgical collateralization provides protection against future infarctions) and patients with less complex disease have equal recommendations for CABG and PCI (where infarction risk is lower and treatment effects may not overcome invasive adverse events).
The LM review6 specifically stresses the need for individual decision making for every patient with LM disease. This is reasonable because the spectrum of patients is wide ranging from patients presenting with isolated ostial lesions and no other CAD to patients with LM disease and massive affections of all other coronary vessels32. Since the occlusion of the LM is most often a lethal event, it is mechanistically plausible to eliminate the need for SYNTAX score calculation. The LM review has also downgraded PCI from a class I to a class IIa recommendation, which finds support in the available data but may also be explained by the association of LM disease with 3-vessel disease in the majority of cases32, 35. However, the review at the same time suggests (in a Table) that patients with proximal and shaft lesions in the absence of significant peripheral CAD should be preferably offered PCI (for reasons of lesser invasiveness) while distal left main disease and more complex CAD should receive a primary recommendation for CABG.
The remaining recommendations from the 2018 Guidelines5 also pragmatically reflect the mechanistic concept. It is important to realize, that exploiting mechanisms requires their continuous presence. That means that bypass grafts and stents alike have to stay patent and complication rates have to be low. In theory, grafting or stenting all vessels with CAD may solve the problem, but in practice, graft failure and stent-related complications force us to be more pragmatic, both on the PCI and the CABG side.
In summary, the current guidelines and the most recent review of the LM section are a valid and plausible reflection of the currently available data in the literature and find ample support from a mechanistic perspective. This mechanistic thinking may provide treating physician with a better understanding of “why” invasive treatment options may be needed for the care of their patients, which should help for individual decision-making, as also emphasized by the guidelines.
1. Sinek S. Start with Why: How Great Leaders Inspire Everyone to Take Action.
2. Doenst T, Schlensak C, Schibilsky D, Faerber G. Do We Need Basic Research in Cardiac Surgery? Thorac Cardiovasc Surg. 2018;66:2-6.
3. Knight KL. Study/experimental/research design: much more than statistics. J Athl Train. 2010;45:98-100.
4. Lewallen S, Courtright P. Epidemiology in practice: case-control studies. Community Eye Health. 1998;11:57-58.
5. Neumann FJ, Sousa-Uva M, Ahlsson A, et al. 2018 ESC/EACTS Guidelines on myocardial revascularization. Eur Heart J. 2019;40:87-165.
6. Byrne RA, Fremes S, Capodanno D, et al. 2022 Joint ESC/EACTS review of the 2018 guideline recommendations on the revascularization of left main coronary artery disease in patients at low surgical risk and anatomy suitable for PCI or CABG. Eur J Cardiothorac Surg. 2023;64.
7. Fox KAA, Metra M, Morais J, Atar D. The myth of ‘stable’ coronary artery disease. Nature Reviews Cardiology.2020;17:9-21.
8. Pasierski M, Staromłyński J, Finke J, et al. Clinical Insights to Complete and Incomplete Surgical Revascularization in Atrial Fibrillation and Multivessel Coronary Disease. Front Cardiovasc Med.2022;9:910811.
9. Serruys PW, Morice M-C, Kappetein AP, et al. Percutaneous Coronary Intervention versus Coronary-Artery Bypass Grafting for Severe Coronary Artery Disease. New England Journal of Medicine. 2009;360:961-972.
10. Stone GW, Kappetein AP, Sabik JF, et al. Five-Year Outcomes after PCI or CABG for Left Main Coronary Disease. New England Journal of Medicine. 2019;381:1820-1830.
11. Miller RJH, Bonow RO, Gransar H, et al. Percutaneous or surgical revascularization is associated with survival benefit in stable coronary artery disease. Eur Heart J Cardiovasc Imaging. 2020;21:961-970.
12. Hachamovitch R, Hayes SW, Friedman JD, Cohen I, Berman DS. Comparison of the Short-Term Survival Benefit Associated With Revascularization Compared With Medical Therapy in Patients With No Prior Coronary Artery Disease Undergoing Stress Myocardial Perfusion Single Photon Emission Computed Tomography. Circulation. 2003;107:2900-2907.
13. van Driest FY, van der Geest RJ, Broersen A, et al. Quantification of myocardial ischemia and subtended myocardial mass at adenosine stress cardiac computed tomography: a feasibility study. Int J Cardiovasc Imaging. 2021;37:3313-3322.
14. Shabbir A, Fan L, Fraser G, Cassar MP, Swinburn J. Quantification of Ischemia As a Prognostic Mandate for Coronary Revascularization in Asymptomatic Patients: How Much Is Enough? Crit Pathw Cardiol. 2019;18:98-101.
15. Doenst T, Bonow RO, Bhatt DL, Falk V, Gaudino M. Improving Terminology to Describe Coronary Artery Procedures: JACC Review Topic of the Week. J Am Coll Cardiol. 2021;78:180-188.
16. Nowbar AN, Rajkumar C, Al-Lamee RK, Francis DP. Controversies in revascularisation for stable coronary artery disease. Clin Med (Lond). 2021;21:114-118.
17. Bajaj NS, Patel N, Kalra R, et al. Percutaneous coronary intervention vs. coronary artery bypass grafting for left main revascularization: an updated meta-analysis. European Heart Journal - Quality of Care and Clinical Outcomes. 2017;3:173-182.
18. Maron DJ, Hochman JS, Reynolds HR, et al. Initial Invasive or Conservative Strategy for Stable Coronary Disease. N Engl J Med. 2020;382:1395-1407.
19. Bytyçi I, Morina D, Bytyqi S, Bajraktari G, Henein MY. Percutaneous Coronary Intervention Is Not Superior to Optimal Medical Therapy in Chronic Coronary Syndrome: A Meta-Analysis. J Clin Med. 2023;12.
20. Galli M, Benenati S, Zito A, et al. Revascularization strategies versus optimal medical therapy in chronic coronary syndrome: A network meta-analysis. Int J Cardiol. 2023;370:58-64.
21. Fearon WF, Zimmermann FM, De Bruyne B, et al. Fractional Flow Reserve–Guided PCI as Compared with Coronary Bypass Surgery. New England Journal of Medicine. 2021.
22. Velazquez EJ, Lee KL, Deja MA, et al. Coronary-Artery Bypass Surgery in Patients with Left Ventricular Dysfunction. New England Journal of Medicine. 2011;364:1607-1616.
23. Perera D, Clayton T, O’Kane PD, et al. Percutaneous Revascularization for Ischemic Left Ventricular Dysfunction. New England Journal of Medicine. 2022;387:1351-1360.
24. Doenst T, Bonow RO, Bhatt DL, Falk V, Gaudino M. Why “Myocardial Revascularization” Inadequately Describes the Mechanisms of Percutaneous Coronary Interventions and Coronary Bypass Surgery. Journal of the American College of Cardiology - In press. 2021.
25. Abdallah MS, Wang K, Magnuson EA, et al. Quality of Life After Surgery or DES in Patients With 3-Vessel or Left Main Disease. Journal of the American College of Cardiology. 2017;69:2039-2050.
26. Sabatine MS, Bergmark BA, Murphy SA, et al. Percutaneous coronary intervention with drug-eluting stents versus coronary artery bypass grafting in left main coronary artery disease: an individual patient data meta-analysis. Lancet. 2021;398:2247-2257.
27. Borges N, Kapadia SR, Ellis SG. Unprotected Left Main Coronary Artery Disease: Management in the Post NOBLE and EXCEL Era. Interv Cardiol. 2017;12:92-96.
28. Lee S-W, Lee PH, Ahn J-M, et al. Randomized Trial Evaluating Percutaneous Coronary Intervention for the Treatment of Chronic Total Occlusion. Circulation. 2019;139:1674-1683.
29. Lee PH, Ahn JM, Chang M, et al. Left Main Coronary Artery Disease: Secular Trends in Patient Characteristics, Treatments, and Outcomes. J Am Coll Cardiol. 2016;68:1233-1246.
30. Yusuf S, Zucker D, Peduzzi P, et al. Effect of coronary artery bypass graft surgery on survival: overview of 10-year results from randomised trials by the Coronary Artery Bypass Graft Surgery Trialists Collaboration. Lancet. 1994;344:563-570.
31. Kang SH, Ahn JM, Lee CH, et al. Differential Event Rates and Independent Predictors of Long-Term Major Cardiovascular Events and Death in 5795 Patients With Unprotected Left Main Coronary Artery Disease Treated With Stents, Bypass Surgery, or Medication: Insights From a Large International Multicenter Registry. Circ Cardiovasc Interv. 2017;10.
32. Mohammad MA, Persson J, Buccheri S, et al. Trends in Clinical Practice and Outcomes After Percutaneous Coronary Intervention of Unprotected Left Main Coronary Artery. J Am Heart Assoc. 2022;11:e024040.
33. Morice MC, Serruys PW, Kappetein AP, et al. Five-year outcomes in patients with left main disease treated with either percutaneous coronary intervention or coronary artery bypass grafting in the synergy between percutaneous coronary intervention with taxus and cardiac surgery trial. Circulation. 2014;129:2388-2394.
34. De Filippo O, Di Franco A, Boretto P, et al. Percutaneous coronary intervention versus coronary artery surgery for left main disease according to lesion site: A meta-analysis. J Thorac Cardiovasc Surg. 2023;166:120-132.e111.
35. Persson J, Yan J, Angerås O, et al. PCI or CABG for left main coronary artery disease: the SWEDEHEART registry. Eur Heart J. 2023;44:2833-2842.
Nothing to disclose
The information and views presented on Latamlaces.org represent the views of the authors and contributors of the material and not necessarily of LACES.
